How Are Pills and Tablets Marked in Medicine?
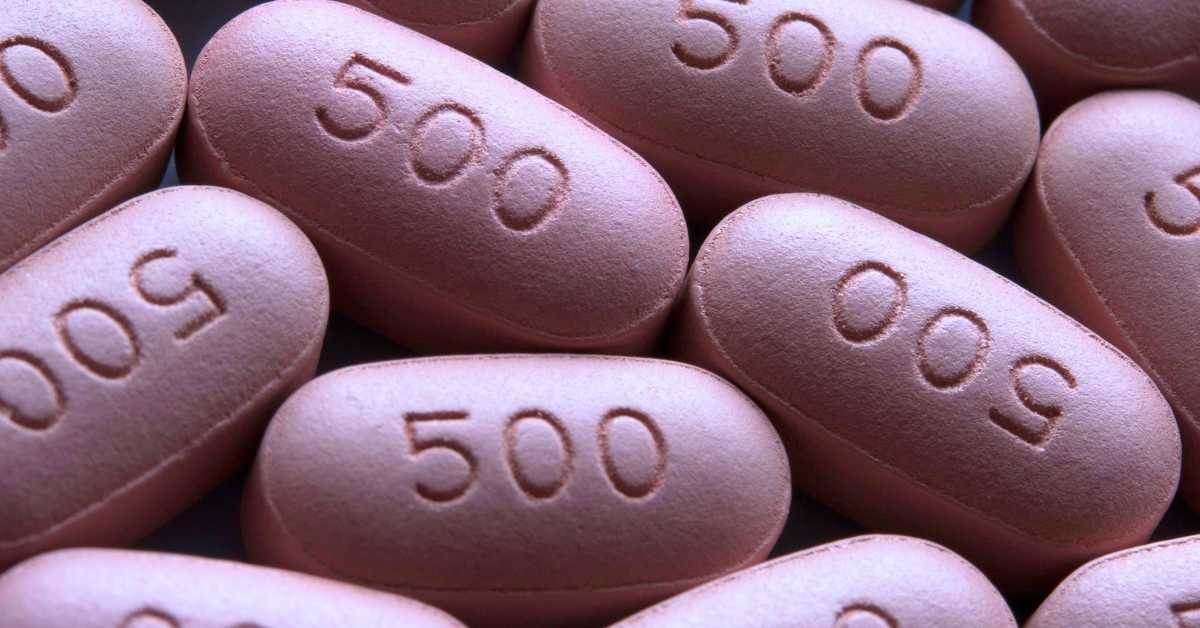
Have you ever wondered how they make those incredibly small but detailed imprints on oral medication? We explain how pills and tablets are marked in medicine and what those markings mean below!
Why Are Pills & Tablets Marked?
Before we get into how drug manufacturers label pills and tablets, we’ll explain why drugmakers must mark them. Pills and tablet markings are required by the Food and Drug Administration (FDA) for consumer safety and are part of the pharmaceutical imprint code.
The pharmaceutical imprint code is a series of letters, symbols, or marks printed, etched, or engraved onto oral medication for identification. All tablets, capsules, and pills have a unique marking archived in the FDA’s national drug code directory that anyone can use to identify medicine. In the directory, people can find the imprint along with the name of the drug, manufacturer, active ingredients, and dosage strength.
How Are Pills & Tablets Marked?
Now you know why, but how do drug manufacturers make such detailed and important marks on pills and tablets in medicine? Laser marking is the most common and effective way to mark oral medication.
Depending on the surface material and the mark, different laser types are used for pill marking in pharmaceuticals, like UV and green lasers. But with advanced laser technology, drug manufacturers can make precise markings, engravings, and etchings on these minuscule and fragile pills and tablets without damaging the medication.
What if a Pill or Tablet Doesn’t Have a Marking?
What should a person do if they come across a pill or tablet that doesn’t have a marking of any kind? If you’re about to take an oral medication and see that it’s blank, it’s likely because it is not FDA-approved, and you should refrain from consuming it.
Vitamins, energy pills, and diet pills do not need to follow the pharmaceutical imprint code, so if that’s the oral supplement you’re taking, you don’t have to worry about the absence of marking. Also, drugs from other countries don’t have to follow FDA-approved guidelines if you purchase medication abroad. A blank pill or tablet could also mean the drug is illicit or a counterfeit, so don’t take the chance, and consider reporting the medication to the FDA’s MedWatch resources.
Now you should understand more about how and why there are small markings and labels on oral medication. Remember, if you’re unsure about a pill or tablet, confirm its identity and safety via its imprint code before consuming it.





